Abstract
Background: Extramedullary B-cell acute lymphoblastic leukemia (EM-ALL) is a rare occurrence characterized by dismal outcome and no defined therapeutic approach. The monoclonal antibody inotuzumab ozogamicin (InO) is approved for treatment of CD22+ B-ALL.
Aims: To characterize a series of relapsed/refractory (r/r) adult EM-ALL patients (pts) and evaluate outcome after treatment with InO.
Methods: We studied 31 r/r pts (median age, 31 years; range, 19-81 years), who were treated with InO between 2015 and 2021 within a compassionate use program (n=7) or on-label after FDA or EMA approval (n=24) All pts were CD22 positive at relapse/progressive disease. Up to 6 InO cycles (≤2 cycles, n=19; 3-4 cycles, n=7; 5-6 cycles, n=5) were administered according to the previously approved regimen. EM response assessment was performed by CT or PET-CT. Prior therapy consisted of intensive chemotherapy +/- tyrosine kinase inhibitors. Allogeneic hematopoietic stem cell transplantation (allo-SCT) was performed in 18 pts (first line or at relapse, n=9, each). Prior to InO, blinatumomab was administered in n=14 and local irradiation in n=5 pts.
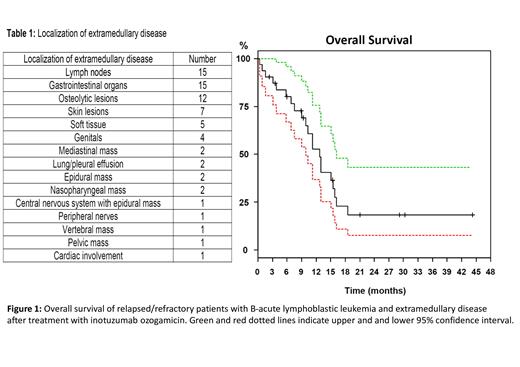
Results: Overall, pts had in median 2 EM manifestations (range, 1-9). Localization of EM disease is shown in Table 1. In addition to EM disease, n=16 (52%) pts had a relapse in bone marrow. At the time of r/r EM-ALL median white blood cell and platelet counts were 5.1/nl (range, 0.04-24.7/nl) and 110.5/nl (range, 6-337/nl), respectively. Fifteen pts (48%) were female; ECOG was ≤ 2 in 29 pts and 3 in 2 pts. Cytogenetic analysis at the time of r/r EM-ALL was available in 13 (42%) pts. Of those, 6 pts had a normal karyotype, 4 were complex, 2 pts displayed a t(9;22) and 1 had an additional X-chromosome.
Seven (23%) of the 31 pts had no response assessment after the first induction cycle including 1 patient who died at day 11 of the first InO cycle due to cerebral hemorrhage. Complete remission assessed by PET-CT (CR; including EM and hematological/bone marrow CR) after the first InO cycle was achieved in 10 of 24 assessed pts (42%), 9 pts (37.5%) had a partial remission (PR), 2 (8%) had stable disease (SD) and 3 (12.5%) showed resistant/progressive disease (RD/PD). After 2 InO cycles, CR was achieved in 17 of 31 pts (55%), PR in 9 (29%); 1 patient (3%) experienced early death and 4 pts with SD+RD/PD did not receive further InO treatment (13%).
Median follow-up was 29 months (95%-CI, 21 months - not reached) and median overall survival (OS) 12.8 months (95%-CI, 9.9-16.2 months; Figure 1). One-year and 2-years OS rates were 53% (95%-CI, 37-76%) and 18% (95%-CI, 8-43%), respectively. In Cox regression analysis age as a continuous variable had no impact on OS (P=0.83). This was also true when using 60 years as cut-off (P=0.2).
Twelve pts went on to allo-SCT (CR, n=6; PR, n=3; PD, n=3). Prior to allo-SCT 8 pts received ≤2 InO cycles and 4 pts ≤4 cycles. Sinusoidal obstruction syndrome (SOS) was reported in 1 patient after transplant; conditioning in this patient consisted of treosulfan/fludarabine/thiotepa. In pts achieving a CR after InO treatment (n=16), median OS was 10 months with no difference (P=0.80) in relapse-free survival (RFS) if an allo-SCT was performed (n=6) or not (n=10). There was no difference on OS (P=0.08) or RFS (P=0.2) if pts had EM manifestations only as compared to EM disease and bone marrow involvement.
In patients with CR/PR after InO treatment, relapse occurred in 10 of 26 pts (38%; after allo-SCT, n=3); of those, all except than one succumbed of their disease. Two pts died in remission (sepsis, SOS/multi-organ failure, n=1; each). One patient experienced a molecular relapse, which could be successfully treated with InO again. Ten pts are in ongoing CR (n=9) or PR (n=1), including the patient with prior molecular relapse and InO re-exposure.
Conclusions: This outcome analysis demonstrates that treatment with InO is an effective and promising approach in r/r-ALL patients with EM disease. However, allo-SCT alone seems not to be effective in maintaining disease control. Thus, autologous chimeric antigen receptor T-cells or advanced bi-specific antibodies as consolidation therapy should be evaluated in the future.
Webster: Pfizer: Consultancy; AmGen: Consultancy. Brunner: Keros Therapeutics: Consultancy; GSK: Research Funding; AstraZeneca: Research Funding; Aprea: Research Funding; Agios: Consultancy; Acceleron: Consultancy; Janssen: Research Funding; Takeda: Consultancy, Research Funding; BMS/Celgene: Consultancy, Research Funding; Novartis: Consultancy, Research Funding. Levis: AbbVie: Consultancy, Honoraria, Membership on an entity's Board of Directors or advisory committees; Amgen, Astellas Pharma, Daiichi-Sankyo, FujiFilm, and Menarini: Honoraria; Pfizer: Consultancy, Honoraria; Takeda: Honoraria; BMS: Consultancy, Honoraria, Membership on an entity's Board of Directors or advisory committees; Jazz: Consultancy, Honoraria; Astellas and FujiFilm: Research Funding. Schlenk: Agios: Honoraria; Astellas: Honoraria, Research Funding, Speakers Bureau; Celgene: Honoraria; Daiichi Sankyo: Honoraria, Research Funding; Hexal: Honoraria; Neovio Biotech: Honoraria; Novartis: Honoraria; Pfizer: Honoraria, Research Funding, Speakers Bureau; Roche: Honoraria, Research Funding; AstraZeneca: Research Funding; Boehringer Ingelheim: Research Funding; Abbvie: Honoraria. Papayannidis: Pfizer, Amgen, Novartis: Honoraria.